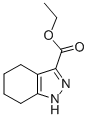
ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate
ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate
CAS: 4492-02-8
Molecular Formula: C10H14N2O2
ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate - Names and Identifiers
ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate - Physico-chemical Properties
| Molecular Formula | C10H14N2O2 |
| Molar Mass | 194.23 |
| Density | 1?+-.0.06 g/cm3(Predicted) |
| Melting Point | 88 °C(Solv: ligroine (8032-32-4)) |
| Boling Point | 403.5±45.0 °C(Predicted) |
| pKa | 12.36±0.20(Predicted) |
| Storage Condition | 2-8°C |
ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate - Introduction
Ethyl 4,5,6, is an organic compound whose molecular formula is C12H15NO2. The following is a description of the properties, uses, preparation and safety information of this compound:
Nature:
-Appearance: ethyl 4,5,6, colorless to yellow liquid.
-Melting point: approximately -70°C to -65°C.
-Boiling point: about 280 ℃ to 285 ℃.
-Density: about 1.11 to 1.13g/cm³.
-Soluble in some organic solvents at room temperature, such as ethanol and dimethylformamide.
Use:
- ethyl 4,5,6, can be used as an intermediate in organic synthesis and for the synthesis of other organic compounds.
-It can also be used in the pharmaceutical field, for example, as a synthetic raw material for antiepileptic drugs.
Preparation Method:
The preparation of ethyl 4,5,6, is usually carried out by the following steps:
1. First, ethyl 2-aminobenzoate is condensed with chloroacetic acid to produce methyl 2-chloroacetate.
2. Then, 2-chloroacetic acid benzene methyl ester is condensed with benzoyl chloride by an oxylic reagent (such as red phosphorus or trimethyl sulfoxide) to generate 4,5, 6,7-tetrahydro-1H-indazole-3-carboxylic acid chloride.
3. Finally, 4,5,6,7-tetrahydro-1H-indazole-3-carboxylic acid chloride is reacted with ethanol to generate ethyl 4,5,6, Ni.
Safety Information:
- ethyl 4,5,6, specific toxicity and safety measures have not been reported in detail. However, since it is an organic compound, it should generally be used and handled under appropriate chemical laboratory conditions.
-Use following proper laboratory procedures and wearing protective equipment such as laboratory gloves and goggles.
-It should be stored in a closed container, away from fire sources and high heat, and avoid contact with oxidants and acids.
Nature:
-Appearance: ethyl 4,5,6, colorless to yellow liquid.
-Melting point: approximately -70°C to -65°C.
-Boiling point: about 280 ℃ to 285 ℃.
-Density: about 1.11 to 1.13g/cm³.
-Soluble in some organic solvents at room temperature, such as ethanol and dimethylformamide.
Use:
- ethyl 4,5,6, can be used as an intermediate in organic synthesis and for the synthesis of other organic compounds.
-It can also be used in the pharmaceutical field, for example, as a synthetic raw material for antiepileptic drugs.
Preparation Method:
The preparation of ethyl 4,5,6, is usually carried out by the following steps:
1. First, ethyl 2-aminobenzoate is condensed with chloroacetic acid to produce methyl 2-chloroacetate.
2. Then, 2-chloroacetic acid benzene methyl ester is condensed with benzoyl chloride by an oxylic reagent (such as red phosphorus or trimethyl sulfoxide) to generate 4,5, 6,7-tetrahydro-1H-indazole-3-carboxylic acid chloride.
3. Finally, 4,5,6,7-tetrahydro-1H-indazole-3-carboxylic acid chloride is reacted with ethanol to generate ethyl 4,5,6, Ni.
Safety Information:
- ethyl 4,5,6, specific toxicity and safety measures have not been reported in detail. However, since it is an organic compound, it should generally be used and handled under appropriate chemical laboratory conditions.
-Use following proper laboratory procedures and wearing protective equipment such as laboratory gloves and goggles.
-It should be stored in a closed container, away from fire sources and high heat, and avoid contact with oxidants and acids.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: Ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate Visit Supplier Webpage Request for quotationCAS: 4492-02-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate Request for quotation
CAS: 4492-02-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 4492-02-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4,5,6,7-TETRAHYDRO-1H-INDAZOLE-3-CARBOXYLIC ACID ETHYL ESTER Visit Supplier Webpage Request for quotation
CAS: 4492-02-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 4492-02-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate Visit Supplier Webpage Request for quotationCAS: 4492-02-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ethyl 4,5,6,7-tetrahydro-1H-indazole-3-carboxylate Request for quotation
CAS: 4492-02-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 4492-02-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4,5,6,7-TETRAHYDRO-1H-INDAZOLE-3-CARBOXYLIC ACID ETHYL ESTER Visit Supplier Webpage Request for quotation
CAS: 4492-02-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 4492-02-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


